Project Feasibility
The bridge between "What if?" and "What’s next?" Every great facility begins with a bold idea. But in high-stakes sectors like Pharma, Data Centers, and Renewable Energy, an idea is only as strong as its foundation. The transition from a concept to a physical asset is where most risks live. A minor oversight in the planning phase can quickly spiral into a massive bottleneck during construction, draining resources and delaying your time-to-market. We specialize in helping you look around these corners, identifying regulatory risks and technical hurdles before they become expensive problems.
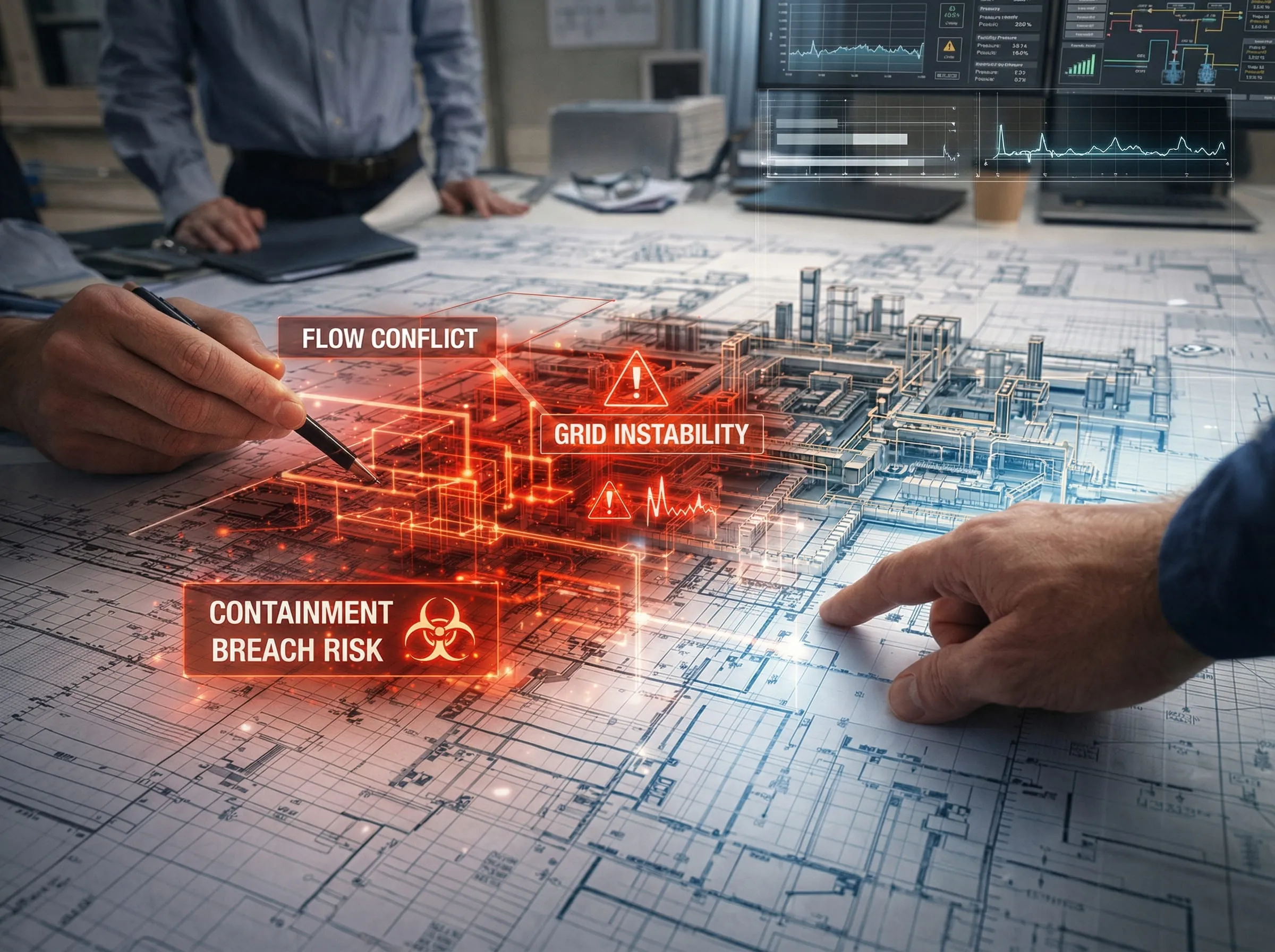
Pre-empting the "Fatal Flaw"
The most expensive mistakes in industrial construction are those that were preventable during the conceptual phase. Technical bottlenecks such as grid-synchronization issues in a Solar farm or flow-path errors in an OEB 6 containment lab, rarely reveal themselves until you are deep into the CAPEX phase. We treat feasibility as a stress test, interrogating every engineering and regulatory variable to catch these obstacles while they are still just lines on a drawing. This proactive approach ensures that once you break ground, your path to commissioning is clear and predictable.